Neural Newsletter for the Week of August 8!
🧠How Thinking Hard Affects the Brain
Everyone knows the feeling of total exhaustion after hammering on a challenging project or slogging through a grueling day at the office. We also may feel as though our willpower and decision-making skills are sapped by these tough periods of thinking, though we may not understand how or why this occurs. So what is the physical basis for the feeling of mental exhaustion? Why is it that our brain feels fatigued after performing certain cognitive tasks, and how does this affect our decision-making?
Researchers from the Paris Brain Institute tackled these questions by using magnetic resonance spectroscopy to measure the brain metabolisms of 40 subjects who were set to perform either easy or difficult cognitive tasks over the period of a typical workday. Difficult, or “high-demand” cognitive tasks, were any activities that could not be ascribed to largely automatic routines, while “low-demand” tasks were more habitual and effortless. An overcommital to high-demand tasks has been shown to generate fatigue and impaired cognitive function and has even been linked to mental health issues like depression and burn-out, but the physical mechanisms behind it were previously unclear.
Early experimentation showed that tougher tasks produced different outcomes in subjects than easier tasks. The researchers write that “choice-related fatigue markers were only present in the high-demand group, with a reduction of pupil dilation during decision-making and a preference shift toward short-delay and little-effort options,” or what the team called a “low-cost bias.” In other words, the subjects that had been assigned the more demanding tasks showed physical signs of cognitive fatigue, i.e. pupil dilation, as well as a behavioral shift away from further mental exertion. The real revelation of the study, however, is the link it establishes between these fatigue indicators and the buildup of toxic waste products in the brain.
They found that their behavioral measures of fatigue correlated with an 8% higher concentration of glutamate in the lateral prefrontal cortex, a brain region associated with cognitive control. When glutamate accumulates extracellularly it can be toxic to the brain, which may tell us something about why we generate the uncomfortable feeling of mental fatigue if we have been under too much cognitive demand.
Unfortunately, since glutamate is the most abundant neurotransmitter in the brain, it would be difficult to develop a pharmacological tool to remedy cognitive fatigue. However, understanding both the biological and behavioral bases of mental effort can help us make more informed decisions about our work-rest habits, and may lead to therapeutic interventions somewhere down the line.
Article:
“A neuro-metabolic account of why daylong cognitive work alters the control of economic decisions” (8/11/2022) Wiehler A, Branzoli F, Adanyeguh I, Mochel F, Pessiglione M. A neuro-metabolic account of why daylong cognitive work alters the control of economic decisions. Curr Biol. 2022 Aug 4:S0960-9822(22)01111-3. doi: 10.1016/j.cub.2022.07.010. Epub ahead of print. PMID: 35961314.
🧠Do our Brains Lose their Sex Differences as we Age?
Examining the biological roots of sexual differences in cognition has become something of a controversial practice, but it’s well established that male and female brains show major differences in gene expression that can impact behavior. However, recent research has revealed that our brains may start losing their sexual differences as we age, with male brains becoming more feminine and female brains becoming more masculine as we grow older. Why is it that our brains appear to get more androgynous as we progress in age?
A Linköping University team based their research question on established evolutionary principles. As organisms enter a weakened state, whether it be due to disease, injury, or a lack of resources, they tend to invest less of their biological capital into the sexual characteristics that would aid immediate reproduction and instead focus more energy on survival and maintenance to reproduce in the future. However, even though older organisms are certainly in a weakened state compared to their younger counterparts, it is unclear whether they would follow this survival strategy. Indeed, since the harmful effects of aging are generally irreversible, an elderly organism may be better served by investing heavily into sexual characteristics and immediate reproduction to quickly pass on its genes before its inevitable death.
To examine which evolutionary path animals tend to follow, the team measured changes in sex-based gene expression in the brains of old and young fruit flies of both sexes. This called for an analysis of the “sexual brain transcriptome” (SBT), which refers to the set of genes in the brain which are expressed more frequently in one of the sexes. A desexualization of the SBT would mean that there is a reduction in the genes that make one sex distinct from the other, and also that there is a drop off in sexually motivated behaviors like mate-seeking and reproduction.
Indeed, after comparing the RNA sequencing of flies that were 5 days old to ones that were 25 days old, the team found that the SBT of the older flies had been significantly desexualized. In the words of the research team, “in both sexes expression of male- and female-biased genes show desexualization as they converge toward an intermediate sexual phenotype with age.” They found that this trend follows a linear relationship - the more a gene is biased towards one sex or the other, the more it loses expressibility as the organism ages. Interestingly, male flies showed a greater drop-off in the expression of sexually biased genes as they got older, meaning that the male SBT appears more sensitive to aging than the female SBT.
Though the team only examined gene expression in the fruit fly brain, they suggest that this pattern of desexualization may apply to other types of species and bodily tissues. Disappointingly, they were unable to find the biological mechanisms that regulated the genetic shift towards desexualization, but this leaves open an exciting opportunity for further research in the near future.
Article:
“Ageing desexualizes the Drosophila brain transcriptome”(8/10/22) - Malacrinò A, Brengdahl MI, Kimber CM, Mital A, Shenoi VN, Mirabello C, Friberg U. Ageing desexualizes the Drosophila brain transcriptome. Proc Biol Sci. 2022 Aug 10;289(1980):20221115. doi: 10.1098/rspb.2022.1115. Epub 2022 Aug 10. PMID: 35946149; PMCID: PMC9364003.
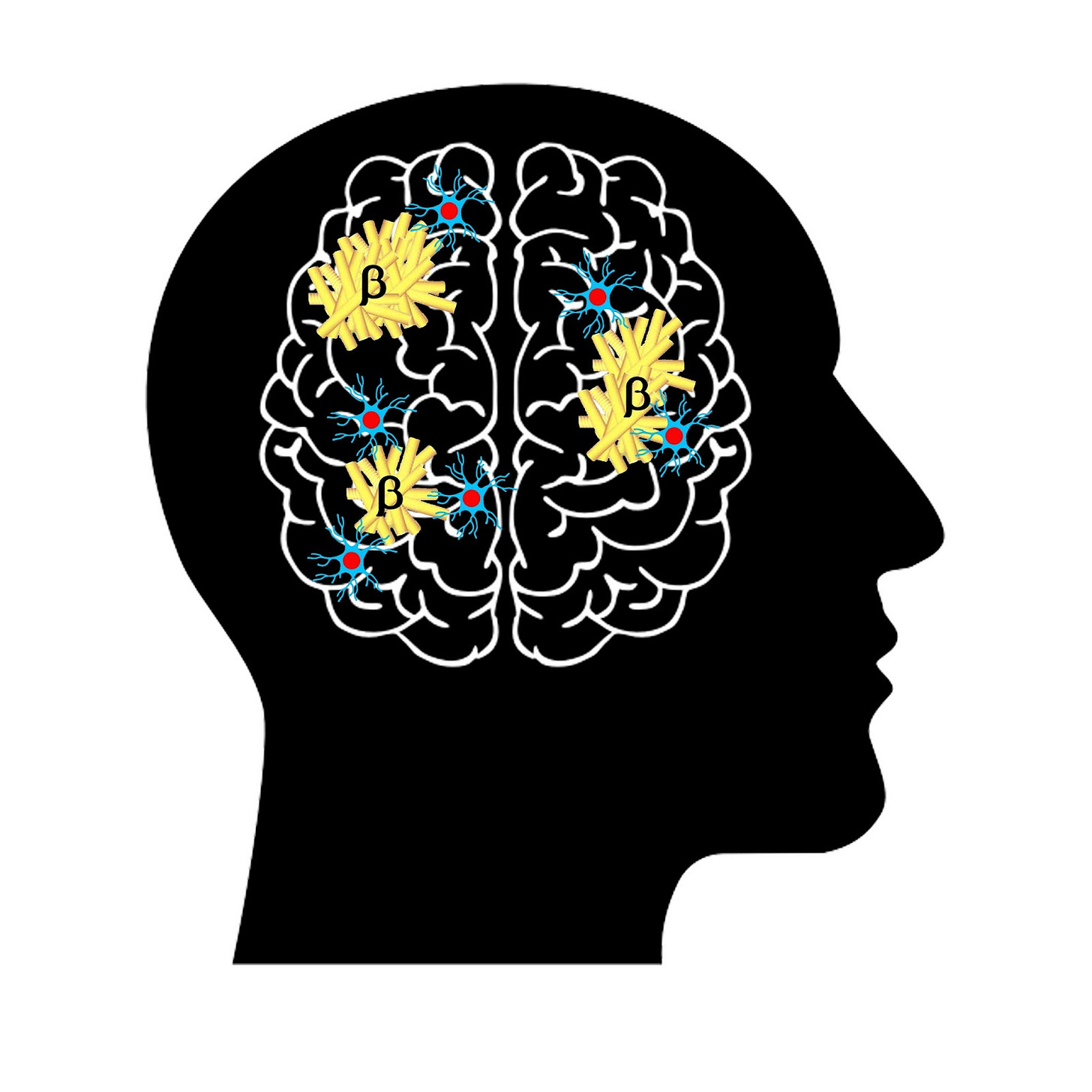
🧠Reprogramming the Brain to Attack Alzheimer’s
Microglia cells are the main warriors in the brain’s immune system, engulfing waste and debris in a cleansing process called phagocytosis. However, these cells have been known to malfunction and contribute to a host of neurological diseases, including Alzheimer’s and other forms of dementia. What makes microglia go haywire, and what lifesaving scientific techniques might be able to put them back on track?
Despite their small size, microglial cells occupy a range of distinct and important roles in the human brain. Some of these roles can be pathological and contribute to disease, but the underlying causes and mechanisms of this behavior are poorly understood. Researchers from the University of San Francisco thought that CRISPR, a technique that involves manually editing certain DNA segments in targeted cells, might offer some insight into understanding how and why microglia go awry, and that it could even help us control them in a way that combats major neurological diseases.
The team developed a brand new protocol for microglia-specific CRISPR application which involved growing human-like microglia from stem cells and altering their genetic code as desired. Using these novel techniques, researchers were able to identify the regions of DNA that caused microglia to act in a harmful manner, including the PFN1 and INPP5D genes in particular. After identifying which regions were at the root of the problem, the team was able to turn on or off the offending genes to get the cells to operate as intended.
A particularly exciting application of this technology relates to Alzheimer’s, as the team demonstrated the possibility that malfunctioning microglia could be coaxed back on track to clear off the harmful plaques which characterize the disease. This unprecedented level of control over specific genes in the brain’s main immune cell opens the door for groundbreaking therapeutic techniques in the future, prompting the head of the study to deem it a “holy grail.” Though the publication is less than a week old, researchers are already beginning to apply these findings to other pharmaceutical interventions to fight back against cognitive disease.
Article:
“A CRISPRi/a platform in human iPSC-derived microglia uncovers regulators of disease states”(08/11/2022) - Dräger, N.M., Sattler, S.M., Huang, C.TL. et al. A CRISPRi/a platform in human iPSC-derived microglia uncovers regulators of disease states. Nat Neurosci (2022). https://doi.org/10.1038/s41593-022-01131-4